ACS Medicinal Chemistry Letters, 2023.
Squaramide Tethered Clindamycin, Chloroquine, and Mortiamide Hybrids: Design, Synthesis, and Antimalarial Activity
Thomas Tremblay, Catherine Bergeron, Dominic Gagnon, Christopher BÉrubÉ, Normand Voyer, Dave Richard, Denis GiguÈre
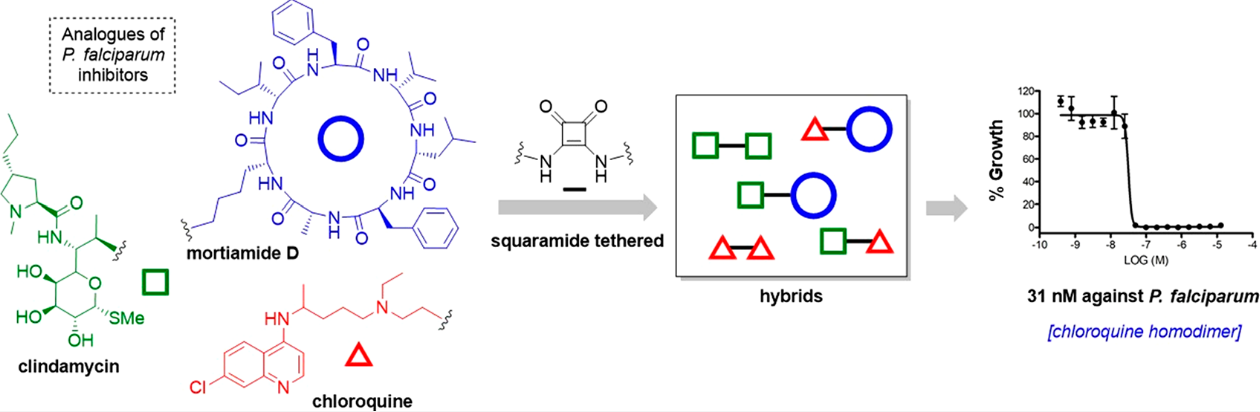
Abstract
Malaria remains one of the major health problems in the world. In this work, a series of squaramide tethered chloroquine, clindamycin, and mortiamide D hybrids have been synthesized to assess their in vitro antiplasmodial activity against 3D7 (chloroquine-sensitive) and Dd2 strains of Plasmodium falciparum. The most active compound, a simple chloroquine analogue, displayed low nanomolar IC50 value against both strains (3 nM for 3D7 strain and 18 nM for Dd2 strain). Moreover, all molecular hybrids incorporating the hydroxychloroquine scaffold showed the most potent activities, exemplified with a chloroquine dimer, IC50 = 31 nM and 81 nM against 3D7 and Dd2 strains, respectively. These results highlight the first time use of clindamycin and mortiamide D as antimalarial molecular hybrids and establish these valuable hits for future optimization.