ACS Omega, 2023.
Determining the Predominant Conformations of Mortiamides A−D in Solution Using NMR Data and Molecular Modeling Tools
Tanos C. C., Franca, Arlan Da Silva Goncalves, Christopher Bérubé, Normand Voyer, Norman Aubry, Steven R. Laplante
Abstract
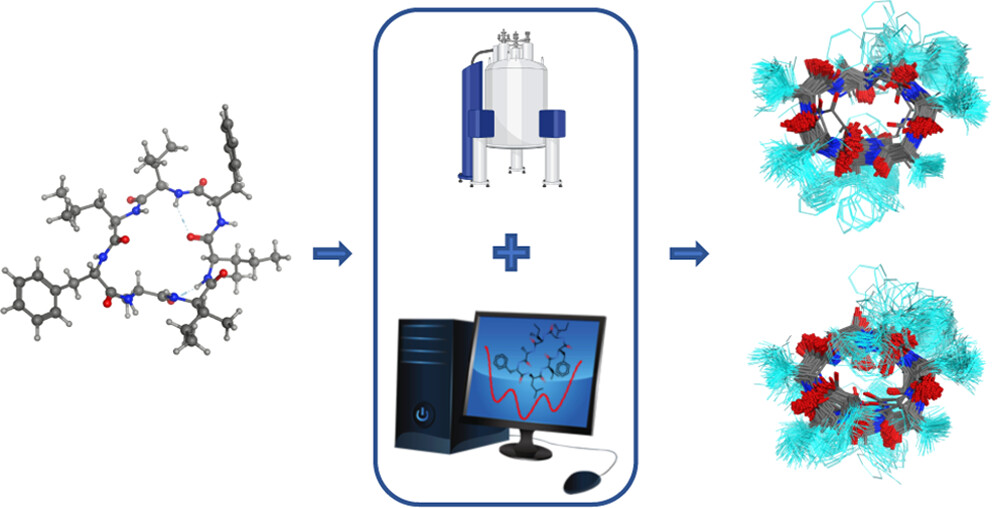
Macrocyclic peptidomimetics have been seriously contributing to our arsenal of drugs to combat diseases. The search for natures discoveries led us to mortiamides AD (found in a novel fungus from Northern Canada), which is a family of cyclic peptides that clearly have demonstrated impressive pharmaceutical potential. This prompted us to learn more about their solution-state properties as these are central for binding to target molecules. Here, we secured and isolated mortiamide D, and then acquired high-resolution nuclear magnetic resonance (NMR) data to learn more about its structure and dynamics attributes. Sets of two-dimensional NMR experiments provided atomic-level (through-bond and through-space) data to confirm the primary structure, and NMR-driven molecular dynamics (MD) simulations suggested that more than one predominant three-dimensional (3D) structure exist in solution. Further steps of MD simulations are consistent with the finding that the backbones of mortiamides AC also have at least two prominent macrocyclic shapes, but the side-chain structures and dynamics differed significantly. Knowledge of these solution properties can be exploited for drug design and discovery.